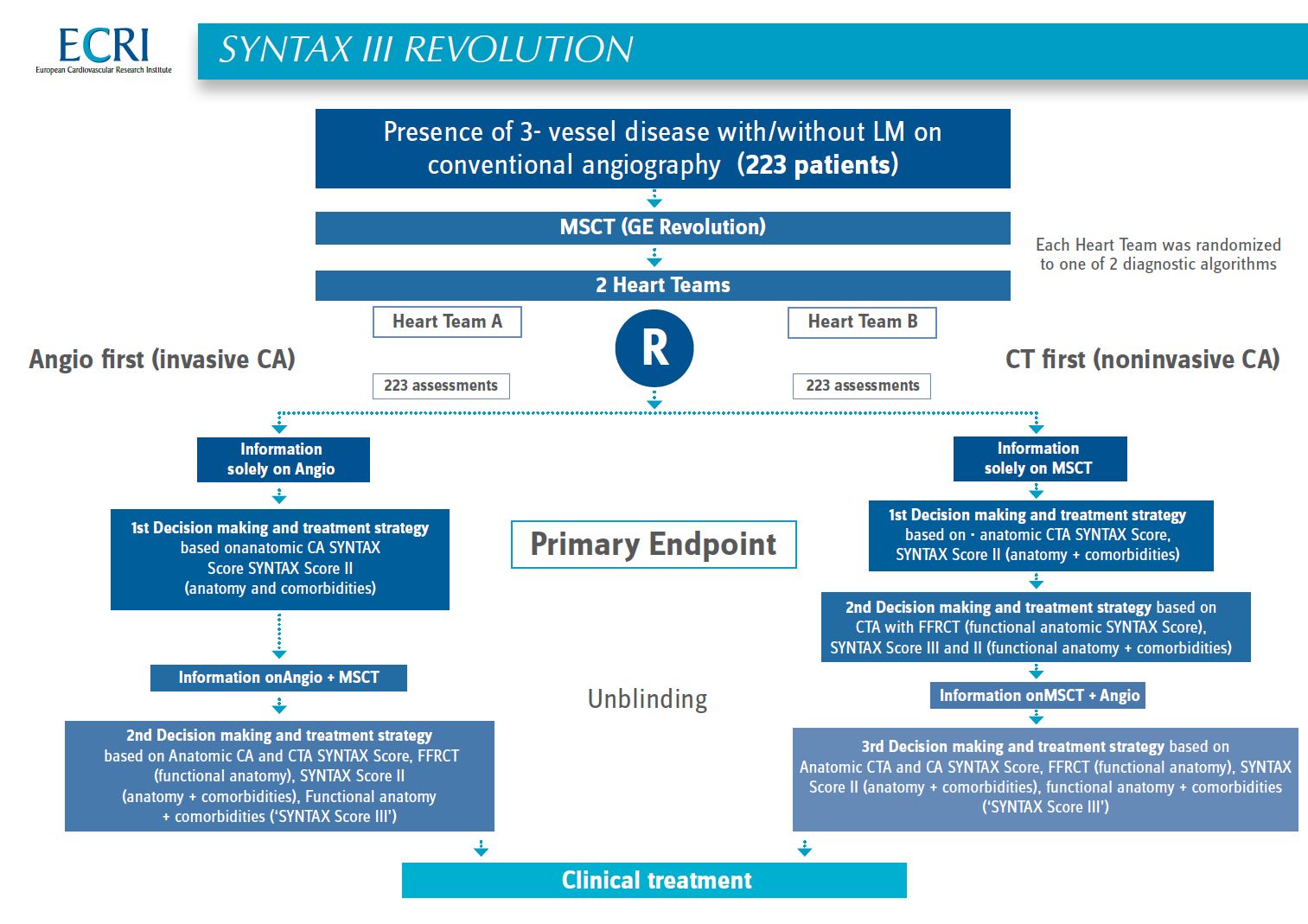
Study design - SYNTAX III Revolution
The SYNTAX III REVOLUTION trial is an investigator initiated, prospective, multicenter, multinational, randomized feasibility clinical trial with an all-comers design. It will randomize two Heart Teams to make a decision between surgical or percutaneous treatment.
The SYNTAX III Revolution trial will be the first study to investigate the role of a decision making process regarding the revascularization strategy based on non-invasive imaging with coronary CTA. In addition, automatic non-invasive assessment of functional coronary anatomy complexity will also be evaluated with the Hearflow algorithm for functional SYNTAX score on MSCT with FFR-CT.
The primary endpoint will evaluate the inter-rater agreement, as assessed by Cohen’s Kappa, on revascularization strategy of two Heart Teams using an “Angio-first” algorithm (based on invasive SYNTAX Score II) or a “CT-first” algorithm (based on non-invasive SYNTAX Score II, without FFRCT).
Patient population:
-Patients with left main (isolated, or associated with 1, 2 or 3 vessel disease) or de novo 3-vessel coronary artery disease (DS ≥50%) who are able to undergo cardiac CT with a GE high-definition RevolutionTM multi-slice CT scanner.
-The enrolment criteria will be unrestrictive all anatomical SYNTAX Scores are eligible for initial screening similar to the SYNTAX I and II studies. As per the original SYNTAX Trial, prior CABG or PCI will be one of the few exclusion criteria.